News
New Publication in the Journal of Catalysis
Phenoxyiminato Cobalt(II) complexes in hydroboration of terminal and internal alkenes
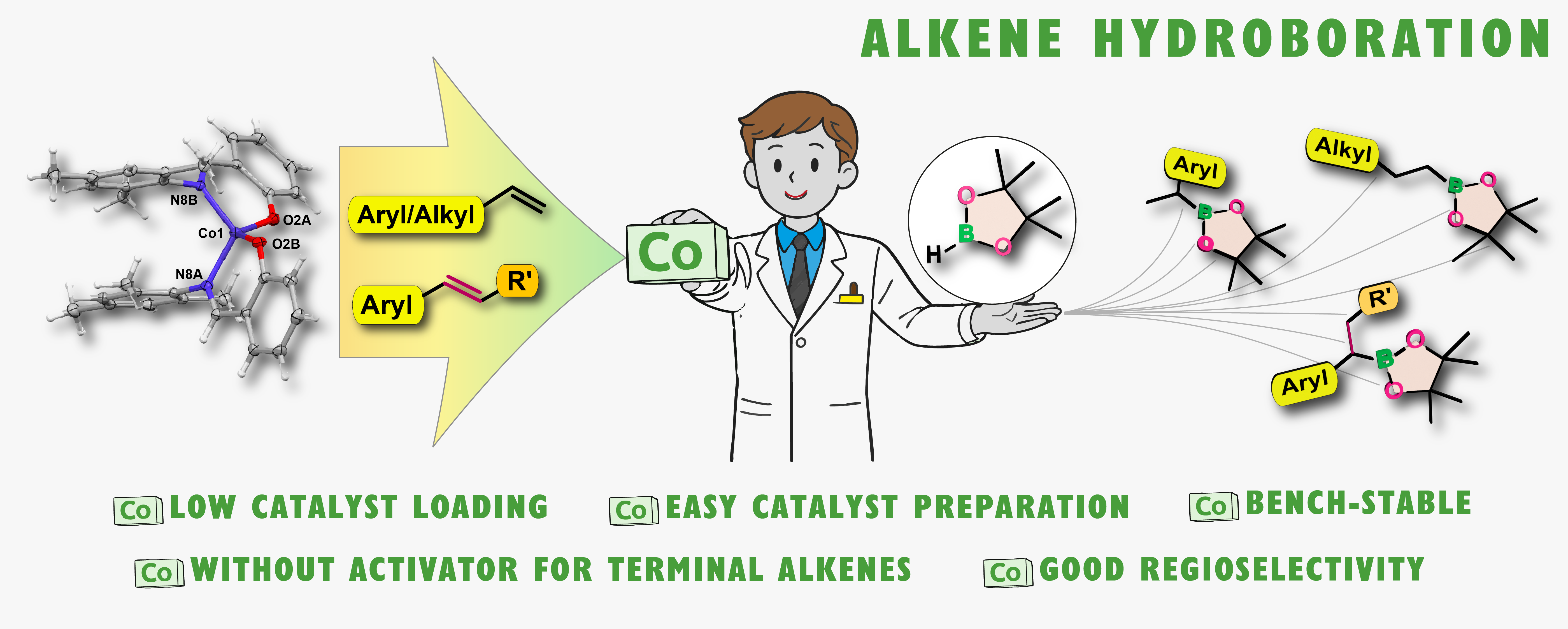
Abstract: Phenoxyiminato cobalt(II) complexes have been developed as efficient catalysts for the hydroboration of alkenes, providing an operationally simple and selective system based on an Earth-abundant metal, offering an alternative to noble metal systems, aiming at more sustainable base metal catalysis. Homoleptic bis(phenoxyiminato)cobalt(II) complexes enable the Markovnikov-selective hydroboration of vinyl arenes with pinacolborane under mild, activator-free conditions at room temperature. In contrast, simple aliphatic terminal alkenes undergo hydroboration with exclusive anti-Markovnikov selectivity. Styrenes with diverse electronic properties as well as functionalised aliphatic alkenes, including vinylsilanes and a vinylphosphonate, were hydroborated with excellent functional-group tolerance and high regioselectivity. Hydroboration of internal alkenes requires modified conditions and proceeds in the presence of a nucleophilic activator, affording moderate to good conversions and revealing the alkene isomerisation processes characteristic of cobalt catalysis. Mechanistic insights into the process were gained using paramagnetic NMR spectroscopy supported by DFT calculations of the reference spectra, which suggest a ligand-assisted activation pathway and the formation of a cobalt hydride as the active form of the complex.
Created on 2026-03-23 by Maciej Zaranek